|
7/29/2023 0 Comments Molecular shapes geometry 
(The lone electron pairs on the O atom are omitted for clarity.) The molecule will not be a perfect equilateral triangle because the C–O double bond is different from the two C–H bonds, but both planar and triangular describe the appropriate approximate shape of this molecule. The three electron groups repel each other to adopt a trigonal planar shape: The central C atom has three electron groups around it because the double bond counts as one electron group. CH 2O has the following Lewis electron dot diagram. A molecule with four electron groups about the central atom, but only one electron group bonded to another atom is linear because there are only two atoms in the molecule.ĭouble or triple bonds count as a single electron group. H 2O is an example of a molecule whose central atom has four electron groups, but only two of them are bonded to surrounding atoms.Īlthough the electron groups are oriented in the shape of a tetrahedron, the shape of the molecule is bent or angular. NH 3 is an example of a molecule whose central atom has four electron groups, but only three of them are bonded to surrounding atoms.Īlthough the electron groups are oriented in the shape of a tetrahedron, from a molecular geometry perspective, the shape of NH 3 is trigonal pyramidal. The straight lines are in the plane of the page, the solid wedged line is coming out of the plane toward the reader, and the dashed wedged line is going out of the plane away from the reader. This diagram of CH 4 illustrates the standard convention of displaying a three-dimensional molecule on a two-dimensional surface. Sulfur hexafluoride is a good example for this. And as we saw earlier in the video, compounds with this molecular geometry are non-polar because there is a symmetric distribution of charges. 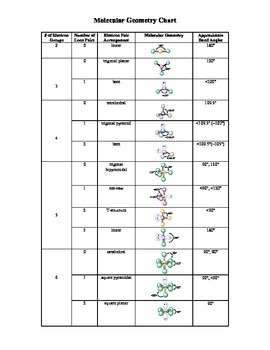
Four electron groups orient themselves in the shape of a tetrahedron. form bonds with identical terminal atoms, the molecular shape of the compound is called octahedral.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
